One of the most important elements of dietary supplement cGMPs is also an item that virtually every brand incorporates into its QA process. Label review is always conducted but not always correctly. The FDA guidance on label review is lengthy and consistently changing. The majority of warning letters from the FDA to brand owners contain some mention of label mistakes, inadequacies, unapproved claims or some other type of label error. The majority of brand owners have one person in charge of label review, often themselves. Large dietary supplement companies dedicate an entire team to the process and incorporate outside counsel when questions arise. The good news is the FDA provides significant and seemingly clear instruction and examples.
The most complete and recent ruling on current labeling requirements for dietary supplements can be found in the Federal Register Vol. 81 No. 103 May 27, 2016p. 33742: https://www.govinfo.gov/content/pkg/FR-2016-05-27/pdf/2016-11867.pdf#page=246
A recent Guidance document was released November 2018: https://www.fda.gov/downloads/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/UCM535372.pdf
With all of this attention, guidance and direction it would seem difficult to make a mistake. A recent review and research of one ingredient, Biotin, showed a different story. The surprising part is the confusion of label guidance with Biotin exists with some of the largest companies in the dietary supplement industry.
The FDA guidance in 2016 and 2018 indicates the Biotin Daily Value established as 30micrograms (mcg) and even provides an example of a correct label: https://www.fda.gov/downloads/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/LabelingNutrition/UCM513884.pdf
However, also found on the FDA website is another document referencing 300mcg as the Biotin daily value: https://www.accessdata.fda.gov/scripts/interactivenutritionfactslabel/factsheets/vitamin_and_mineral_chart.pdf
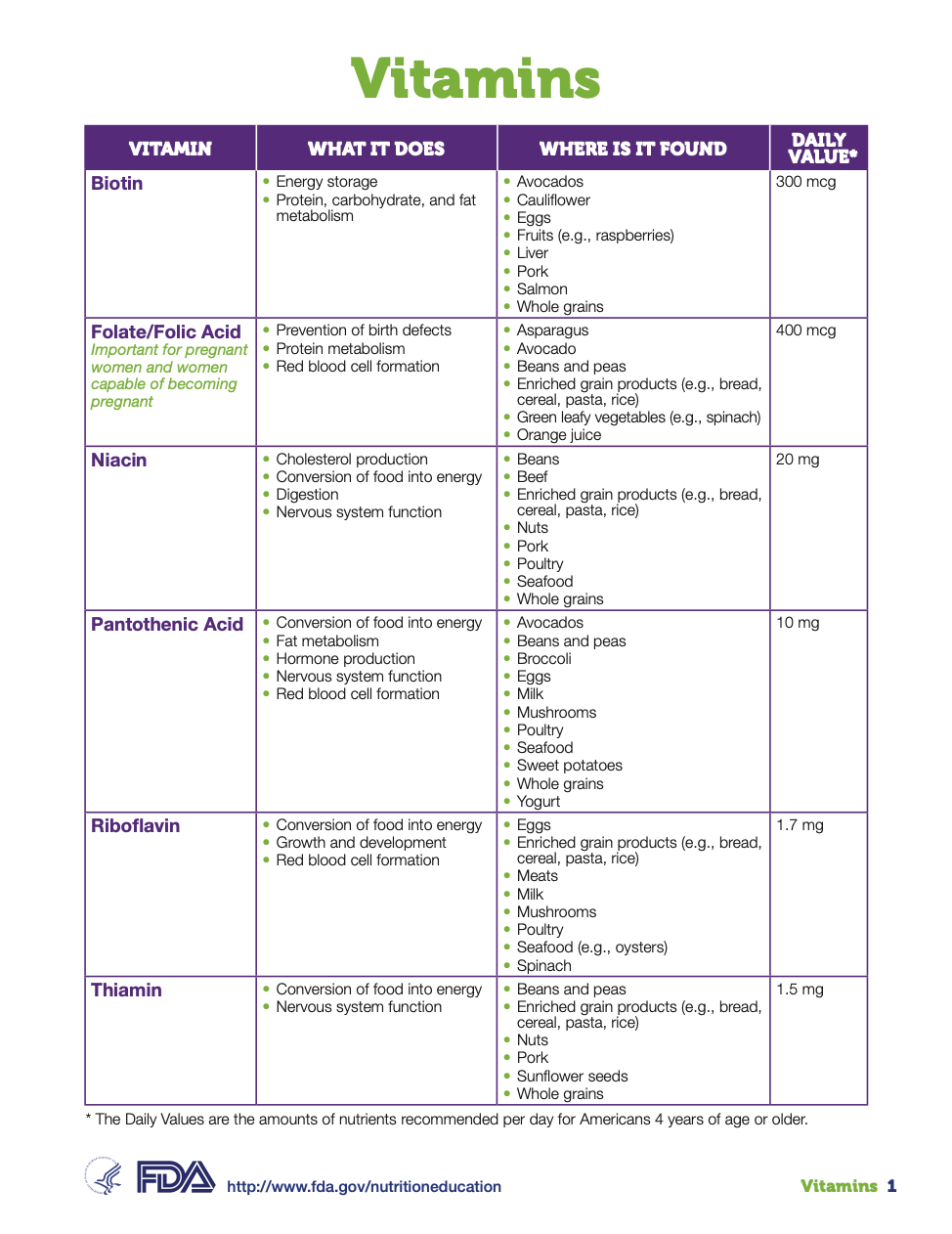
Now look at these 6 products from Doctor’s Best, Natrol, Nature Made, Natures Plus, Amazon Solimo and Amazon Elements:

These six products have 4 different calculations and two different calculations come from the same brand owner, Amazon!
Doctor’s Best confirmed they have changed their Biotin Daily Value to reflect the 30mcg Daily Value and their website confirms that fact. All of these companies are considered sizeable in comparison to the majority of the rest of the industry. Amazon certainly has the manpower and opportunity to at least be consistent in messaging, if not accurate. This example is an indication that the label review process is complicated, changing and an important representation to both the consumer and regulatory environment.
It is important to maintain an awareness of the required changes as well as accuracy to detail. We at Industry Transparency Center are happy to assist with this endeavor.
In good health,
Scott
